INDUSTRIAL TRAINING REPORT
The INDUSTRIAL TRAINING REPORT submitted in fulfillment of the requirement for the
degree of
Bachelors in Pharmacy
By
Ram Singh

University Institute of Pharmaceutical Sciences
Punjab University

University Institute of Pharmaceutical Sciences
Punjab University
Certificate
This is to certify the INDUSTRIAL TRAINING REPORT submitted by my Mr. Ram Singh, Reg. no. 22PY010 to the University Institute of Pharmaceutical Sciences, Punjab University in partial fulfillment of the requirement for the degree of Bachelor in Pharmacy is a bonafide record of work carried out by him under my supervision. The content of this INDUSTRIAL TRAINING REPORT, in full or in part has not been submitted to any other institute or university for the award of any diploma or degree
Internal guide ……
External guide……
ACKNOWLEDGEMENT
It was indeed a great pleasure for me to undertake my one-month summer training at PHARMAHUB, Roorkee. It was a great experience for me as this was my first exposure to the pharmaceutical industry which help me to improve my practical skills.
First and foremost, we would like to thank the Almighty God who enabled me to finish this industrial training and report successfully and above all, for giving me the wisdom and energy to execute the site works.
Our special thanks go towards the personal Mr. B.K. Singh (Plant head), Mr. D.N Jha (G.M. production), Mr. Adwani Yadav (DGM Q.A. ), and Mr. Lala Ram (H.R. Manager).
And I would also thank all the staff members of PHARMAHUB for their kind coordination, guidance, supervision, cooperation, and visual support throughout the tenure of the training. I would like to give special thanks to Mr. Adwani Yadav who help me to prepare the training report.
INTRODUCTION
Pharmahub is one of the leading pharmaceutical companies in India and it has other manufacturing units in different countries like China, US, Russia, UK, Nepal, France and many more.
Pharmahub’s manufacturing facilities in all its plant have the approval of the major regulatory bodies, including the UK’s MHRA(Medical and Health Care Agency) and US FDA(Federal Drug Authority), ANVISA with capabilities for finished dosage formulations.
The output includes:
- Sterile (Injectables)
- Orals (Tablets & Liquids)
- Topicals (Creams & Ointments)
General Requirements :
This company fulfills all the requirements according to WHO, GMP, MHRA & USFDA.
Organization and Personal :
- Assignment of all units of total responsibilities for ensuring that Procedure exit and are followed to ensure product quality.
- Personal qualification persons are qualified by training and experience to perform the work.
Medical facilities:
- First aid facilities are available.
Building & facilities :
Construction features and design according to GMP.
- 5-micron filtered air with temp. 25°c is provided in the liquid section, capsule section and raw material room.
- Well Lighting facilities and all lighting are flashes from the roof.
- Air filtration and cooling are according to suitability to production.
- Double door entry for manufacturing area.
- All doors are constructed with an aluminum frame with glass.
- Windows are constructed with aluminum-coated frames with glass.
- There is no wooden furniture, only stainless steel furniture is used.
Equipments:
Equipments are placed self contain facilities so that cross-contamination is avoided.
- Equipment suitable design, size and exit at suitable location.
- Suitable method and process for easy cleaning and maintenance.
- Filters do not release fibers in the product.
SANITIZATION AND HYGIENIC CONDITION
Sanitization :
- Strict sanitization conditions are maintained throughout the entire plant in order to prevent contamination.
- The product room is kept tiled and scrupulously clear.
- The walls and floors are tiled so it is easy to spray and washed with air antiseptic solution.
- The benches are stained less and still capable of being washed.
- All walls are painted with washable paint.
- All restricted areas are sanitized with insecticide spray every day.
- Water tanks are washed every day.
- All devices are cleaned by a vacuum cleaner.
- Walls and floors are cleaned with wet muff., phenyl muff and then the antiseptic solution.
- Equipments are cleaned according to SOP(Std Operation procedure)
- Antiseptic solution- Teepol – 1% solution
Dettol – 2.5% solution
Savlon – 2.5% solution
Hygienic condition :
- Hygienic conditions are applied to every person who entered the industry.
- All workers should be free from contagious or obnoxious diseases.
- Their clothing is brown in color, which is suitable for the nature of work and climate.
- There are adequate facilities for personal cleaning such as clear towels, soaps, and hand scrubbing brushes provided separately for each sex.
- Separately arranged for labeling and packaging.
- The finished product is stored in a separate cool & dry place.
INDUSTRIAL TRAINING REPORT FLOW CHART
FLOW CHART OF PRODUCTION:
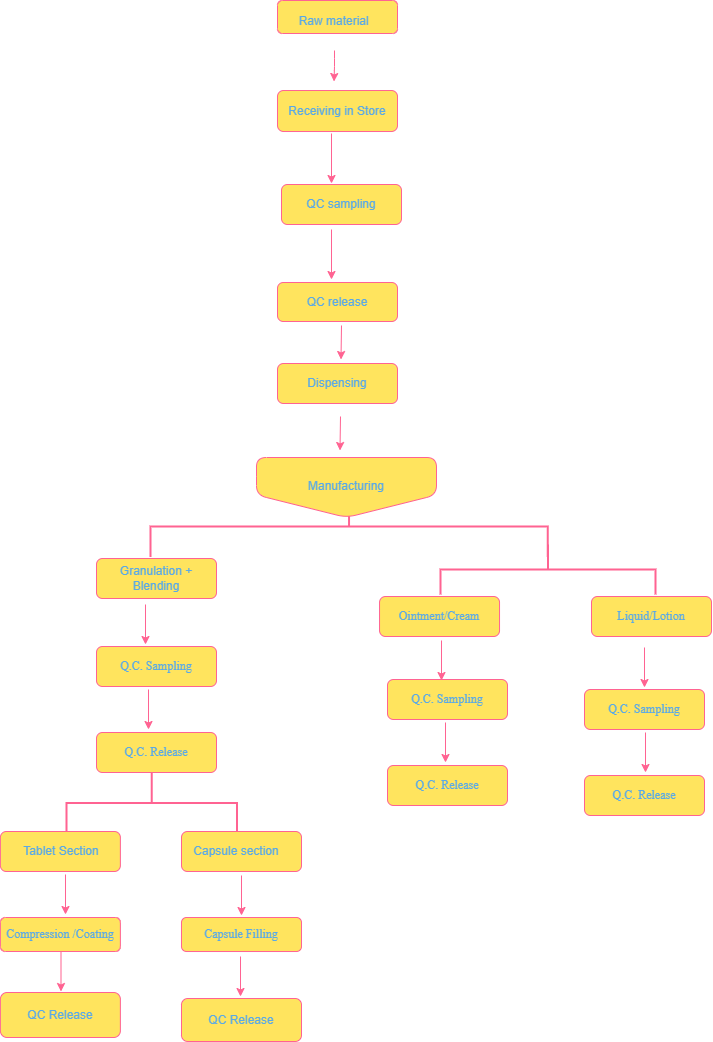

TABLETS & CAPSULES
Tablets are solid unit oral dosage forms containing a medicament or mixture of medicaments and excipients compressed or molded into solid cylinder shape having either flat or concave surfaces. These are the solid dosage forms in which powdered medicament is incorporated.
- Tablets are made by compression of the powder.
- Shape or size may be rounded, oval, elongated etc.
- In many tablets granules are used instead of powders.
- Granule is a generic term used for a small particle or grain.
- Granules are usually obtained by adding liquids(Binders or solvent solutions).
Advantages of Tablets:
- Ease of accurate dosage.
- Physical and chemical stability is good.
Criteria for good tablet:
- Tablets should not be too hard.
- Should be accurate and uniform weight.
- The shape of tablet administration.
- Should not be any manufacturing defects like cracking chipping .
- Should not break during transportation.
- Should disintegrate easily.
- Should be stable chemically and physically.
Preliminary Precautions:
- Good manufacturing practices shall be followed throughout manufacturing process. Ensure area and equipment is clean before commencing manufacturing operation.
- Check that all balances, which are to be used, are calibrated.
- Ensure that Product containers and Equipments are labeled at all stages of Manufacturing.
- Transfer all the dispensed material from the store to the manufacturing area at one time. Counter-check and record the identity and weights of all the ingredients received. Do not use any material, which does not comply in weight or analytical report numbers.
- Maintain and record the environmental conditions of all process areas. The temperature shall not exceed 27°C and relative humidity shall not be more than 50 % during processing and storage of in-process materials.
- Use hand gloves and a mask while handling the product/material.
- Follow the current Standard Operating Procedures.
- Store the material, in-process material, semi-finished and finished material in the designated area properly labeled mentioning the status of the material and batch details.
Manufacturing Process:
Step No. I: Sifting:
Check Sieve Integrity before and after use.
Step No. II: Binder preparation:
– Make a starch paste by mixing starch with hot Purified water IP in a paste kettle at 90°c.
Step No. III: Mixing/Granulation:
Granulation: It is the process in which granules are prepared to consist of solid, dry aggregates of powder particles sufficiently resistant to withstand handling and are intended for oral administration. It is formed by compression or by using a binding agent. The granulation process combines one or more powders and forms granules that will allow the tableting process to be predictable and will produce a quality tablet.
Reason for Granulation :
- To improve powder flow properties.
- To improve compressibility.
- To reduce fines.
- To control the tendency of power to segregate.
- To control Density
Powder flow & Blending :
There are different variables that can contribute to the success or failure of powder, i.e.
- Particle size
- Size distribution
- Cohesivity
- Surface texture
- Surface coating
- Particle interaction
Fluidized Bed Equipment:
- It is a cylindrical body consisting of 8 basic parts.
- Air treatment package
- Product bowl
- Expansion chamber
- Filter system
- Bag house
- Air turbine
- Suspension system
- Control panel
Exclusive features of FBD:
- Continuous production m/c without human touch.
- Suitable for crystalline materials
- Fully automatic-needs little running attention
- Online particle size reduction
- Adequate cleaning access & view window
- Uniform heat transfer
Basic processing cycles:
- Pre-heat
- Spray
- Drying
Step No. IV: Drying:
- It is a very critical step since the moisture level in the granules defines the quality of the tablet.
- As excess moisture level can result in Picking & Sticking to the punch surfaces & also poor flows.
- Overdrying could result in Hardness & Higher friability.
- So, the outlet temperature should be monitored carefully and recorded during drying and the corresponding LOD(Loss On Drying) is recorded.
- The outlet range should be established as per requirement to attain the desired moisture level in granules.
Types of Granulation:
- Dry Granulation
- Wet Granulation
Dry Granulation:
- It is the process used to form Granules without using a liquid solution because the product to be granulated may be sensitive to moisture and heat.
- Forming granules without any moisture requires compacting and densifying the powders.
- A sifting and blending sifter is required to get the required size of material.
- Blender: It is used to mix the material with lubricant.
- The Final material is then weighed and sent to compression and capsulation.
Wet Granulation:
- The process of adding a liquid solution to powders is one of the common ways to granulate.
- Wet granulation is used to improve flow, compressibility, bio-availability and homogeneity of low-dose blends, electrostatic properties of powders and stability of dosage forms.
- As the liquid bridges between the particles are formed, granules are subjected to coalescence alongside some breakage of the bonds.
- The granule size is strongly dependent on the specific surface area of the excipients, as well as the moisture content and liquid saturation of the agglomerate.
- During the wet massing stage, granules may increase in size to a certain degree while the intra-granular porosity goes down.
- However, some heating and evaporation may also take place leading to a subsequent decrease in the mean granule size, especially in small-scale mixers.
Procedure:
Shifting (all materials are sifted in the sifter to decrease the size)
↓
Granulation in RMG(Repaid mixer Granulator)
↓
(Powder + Binder)
↓
FBD
(Fluidized Bed Drier-used for drying in which finger bags to stop material to gone and maintain fluidization)
↓
Sifting (again the material are sifted here)
↓
Milling (Material remains is undergo milling to decrease size)
↓
Sifting (After milling again the material is sifted)
↓
Blending (Material + Blender + Lubricant)
↓
Q.C. Sampling
↓
Q.C. Release
Step No. V: Cutting/milling:
Reducing the particle size of the solid as per BMR.
Step No. VI: Lubrication :
Sift the lubricants & Keep them separately. Load the material in the contagonal blender & blend it. Then add the lubricant and blend it. Collect the weight of granules and affix the status label of conta bin blender.
Step No. VII: Compression :
Conta bin blander having lubricated bland shifted into the compression area. Tablets are prepared different shapes and size according to respective punch and die.
Step No. VII: Coating
The coating is the process of varnishing tablets for their pharmacological function. It is the method to distaste the bitter taste of medicament and produce the desired action at the desired location.
Types of coating:
- Sugar coating
- Film coating
- Enteric coating
- Encapsulation
Sugar coating:
Sugar coating is an effective process for the application of thick coating layers, primarily for masking taste. The syrup is sprayed onto the tablets and dried by air. The thickness of the coating is generally between .5mm to 2mm.
Stages of coating:
- Sealing
- Sub-coating
- Smoothing
- Color coating
- Polishing
- Printing
Traditional sugar coating process:
| Steps | No. of Application | Processing time(min) |
| Sealing | 2-6 | 20-60 |
| Sub-coating | 40-80 | 400-800 |
| Smoothing | 0-10 | 0-100 |
| Color coating | 10-30 | 100-300 |
| Polishing | 2 | 20 |
Advantages of Sugar coating:
- Low-cost material
- Formulation work is easy
- Simple equipment
- Attractive appearance
Disadvantages of Sugar coating:
- Long process time
- Skill dependant process
- Variable quality
- Identification needs extra steps
Film coating:
Film coating is the process whereby a tablet or pellet is surrounded by a thin layer (20-200micron) of polymeric material.
Reasons for film coating:
- Appearance-To changes the color, for branding purposes or other aesthetic reasons.
- Stability- To protect the active ingredient from moisture, light, and the acidic environment of the stomach.
- Taste/Odor- To provide an easy-swallow tablet without the bitter taste of ingredients.
- Release characteristics- Many film coating materials have functional properties which enable the creation of sustained or delayed release dosage forms.
Polymeric materials used:
- Cellulose
→Hydroxypropylmethylcellulose
→Hydroxypropylcellulose
→Ethylcellulose
→methylcellulose
→Carrageen
- Polyethylene Glycol (PEG)
Enteric coating:
The enteric coating is supposed to make the tablet easier on the stomach. It may delay the dissolution of the tablet somewhat but is not generally considered a “slow release”.
The primary reason is to allow the tablet to exit the stomach before it dissolves.
Advantages:
- Time-concurrent with core compression
- Totally dry process
- Formulation flexibility
Disadvantages:
- Equipment cost
- Complex equipment
- Skilled set-up
Encapsulation Advantages:
- Dry process
- Easy formulation
- Formulation flexibility
Disadvantages:
- Natural material
- Complex equipment
- Cost
- Tampering
Tablets & Capsules formed by the company:
- ketoconazole
- Rabeprazole
- Albendazole
- Ranitidine
- Paracetamol
- Citrine
Capsule Filling Machine:
Temperature-25ºc
RH – between 40 to 60
Parts of Filling Machine:
- Short Elevator
- Empty capsule Hopper
- Magazine one by one capsule
- Rectifier Block
- Segments (Eject the capsule in body bush and cap bush)
- Powder Hopper
- Doser form
- Capsule closing then ejection
- DP 100 dusting and polishing of the capsule
- Shorter Plate
- Shorter Roller
- FCE- Filled Capsule Elevator
- ECS-Empty Capsule Shorter
- Metal Detector
- Disintegration Test performed after 2-2 hours
- Length checked of the capsule by Vernier caliper
- Weight- After ½ – ½ hours are checked by analytical balance
Packing of Tablets & Capsules:
- Primary packing: In this the packing is in direct contact with the medicament, e.g. PVC, PVDC, Alu foil etc.
- Secondary packing: In this packing tube is covered with packing like cartoons etc.
- Tertiary packing: in this packing bulk packing is there with the shipper etc.
- OPERATION & CONTROLS:
- Maintain temperature and Relative Humidity of Blister Packing area as Temperature NMT 27°C and RH NMT 55% respectively.
- All equipment and accessories required for use should be cleaned as per the respective Standard Operating Procedure.
- Label all the containers with appropriate labels.
- Check and collect the packaging material from the warehouse and transfer it to the parking area.
- Counter-check the weights of all tablets received and all packing materials received for the batch.
- Duly enter the observations in the BPR.
- Destroy all the leak test samples.
LIQUID MANUFACTURING PROCESS
Preliminary Precautions during liquid Manufacturing:
- Good manufacturing practices shall be followed throughout manufacturing process. Ensure area and equipments are clean before commencing manufacturing operation.
- Check that all balances, which are to be used, are calibrated.
- Ensure that Product containers and Equipments are labeled at all stages of Manufacturing.
- Transfer all the dispensed material from the store to the manufacturing area at one time. Counter-check and record the identity and weights of all the ingredients received.Do not use any material, which does not comply in weight or analytical report numbers.
- Maintain and record the environmental conditions of all process areas. The temperature shall not exceed 27°C and relative humidity shall not be more than 55 % during processing and storage of in-process materials.
- Use hand gloves and a mask while handling the product/material.
- Follow the current Standard Operating Procedures.
Store the material, in-process material, semi-finished and finished
The Facility consists of:
- Sugar syrup vessel
- Online sugar syrup pre-filter
- Manufacturing vessel
- Storage vessel
- Vacuum system for transfer of sugar syrup
- Filter press / Inline Homogenizer
- Product piping
- Control panels
Features of the Facility:
- The plant is designed to be operated by one operator and one helper, saving on manpower costs.
- Sugar syrup and manufacturing vessels are provided with limpet coils for heating and cooling, designed for internal vacuum to facilitate the transfer of sugar directly from the store to the sugar syrup vessel.
- Sugar syrup is transferred to the manufacturing vessel through online sugar syrup. Pre-filtered by vacuum.
- Entry of all propeller agitators is from the bottom through a specially designed cartridge mechanical seal with TC/TC seal.
- The advantage of the bottom propeller agitator over the conventional agitator is that there are no vibrations of the shaft and no couplings in the drive assembly, leading to lower maintenance costs.
- Pipes, pipe fittings and valves are of SS316, seamless, and internally electro-polished with DIN standard unions and silicon Gaskets.
- Manholes of all the vessels are equipped with davit fitting and the cover of the manhole slides instead of being lifted.
Capacities available: 10,000 liters
Manufacturing Process of Liquid :
Step No. I: Clean the Sugar syrup manufacturing tank.
Step No. II: Collect Purified Water in this tank. Add sugar through the charging pump into the Sugar syrup manufacturing tank by operating the tank.
Step No. III: Heat the purified water to approximately 100ºC with continuous stirring and check for complete dissolution. Cool the syrup to a temperature below 40°C by operating the tank.
Step No. IV: Clean the Bulk manufacturing tank.
Step No. V: Add a sufficient quantity of purified water to Step no.IV and to it add active ingredients and mix it properly for at least 3 hours.
Step No. VI: Dissolve the preservatives like Sodium Methyl Paraben, Sodium Propyl Paraben, Sodium Benzoate, Sodium Saccharin & Citric acid in water in a separate S.S container. Transfer the Step VI to Step III. Filter Step III and transfer it to the bulk manufacturing tank.
Step No. VII: Dissolve Aerosil in sufficient water and add to Step III. And then also add Glycerin and Sorbitol to Step no.III with continuous stirring.
Step No. VIII: Transfer the whole solution from the sugar tank to Mfg. tank
Step No. IX : Dissolve Xanthan Gum in warm water & add to step no.-VIII
Step No. X: Add Carboxy Methyl Cellulose to the Mfg. tank & then add Tween-80.
Step No. XI: Dissolve colour in water & add to Step VIII followed by the flavor Sweet Orange.
Step No. XII: Make the final volume to 10000.00 Liters in a bulk manufacturing tank with purified water and continue stirring for another 15-30 minutes.
Check the final pH.
Step No. XIII: Clean Liquid Storage Tank.
Step No. XIV: Transfer & filter the whole suspension to Liquid Storage Tank,
Step No. XV: Inform QA to draw a sample of Formulated bulk. Send Test Request Slip for Analysis to QC through QA Department.
Packing of liquid preparation:
- After filling, bottles are transferred to the packing section.
- In the packing section, the bottles and their caps are checked and labeled.
- The labeled bottles are packed in the folded cartoon and cartoons are also labeled.
- After packing these cartoons are transferred to finished goods storage.
- Precaution
- Maintain Temperature and Relative Humidity of Packing area as Temperature NMT 27°C and RH NMT 55% respectively.
- All equipments and accessories required for use should be cleaned as per the respective Standard Operating Procedure.
- If the wrapper is loose, then check the quantity in Shipper and affix the “Loose Pack” label. Mention the loose quantity on the label. Get the quantity in the box verified by QA Dept.
- Keep packaging material duly labeled and segregated for different products and different batches to avoid intermixing.
- Check and collect the packaging material from the warehouse and transfer it to the packing area.
- Duly enter the observations in the BPR.
- Destroy all the leak test samples.
The major liquid produced:
Ascoril syrup
Paracetamol suspension
Alcal D
OINTMENT SECTION
This department consists of
- Wax heating vessel
- Water melting vessel
- Manufacturing vessel
- Storage vessel
- Interconnecting pipeline
- Pumps (Bump pumps &metering pumps)
- Integrated automatic control panel
- Working platform
Procedure:
- All vessels are manufactured from S.316 grade stainless steel sheets and are GMP-compliant construction.
- The vessels are jacketed, insulated, cladded and with suitable agitator assembly in each vessel.
- Wax is melted in the wax melting vessel.
- Water is heated in a water-heating vessel.
- Both wax and water are transferred into the manufacturing vessel automatically through the vacuum.
- In the Manufacturing vessel both wax & water are homogenized to make a uniform emulsion and cooled by passing chilled water into the jacket of the Manufacturing vessel.
- After the emulsion is formed active ingredients/color etc. are added and thoroughly mixed and homogenized.
- Later necessary perfume is added in case of cosmetics or wherever necessary.
- The ointment/cream is ready.
- The same is transferred by the bump pump into the storage vessel.
- From the storage vessel it is automatically transferred into the filling machine by means of a metering pump.
- The flow rate of the metering pump can be set as per the tube filling machines’ speed and capacity.
Salient Features:
- Complete untouched process, consisting of wax phase, water phase, manufacturing vessel and storage vessel.
- Hydraulic power pack for lid lifting.
- Homogenizer mounted from the bottom
- PLC-based control panel
- Load cell for online weighing
- Lower maintenance cost as no moving components is used.
- Special contra rotary type anchor with blades
- Interconnected pipeline up to the storage tank
- Available from 100 kgs to 500 kgs.
- Also available closed a type dished lid.
Tube Filling, Sealing & Crimping Machine:
- This machine is available in single-head as well as double-head model used for filling, sealing, crimping aluminum, and lami
- It is provided with a “no tube no fill” device all operations are fully automatic except feeding of tubes.
Packing of Ointment preparation:
- After filling the tubes are transferred to the packing section.
- In the packing section, the tubes and their caps are checked and labeled.
- The labeled tubes are packed in the folded cartoons and the cartoons are also labeled.
- After packing these cartoons are transferred in finished goods storage.
Major ointment or creams produced:
- Mupirocin
- Halobetasol
- Candid STABILITY
The stability of a pharmaceutical product may be defined as the capability of a particular formulation in a specific container to remain within physical, chemical, microbiological, therapeutic and toxicological specifications. These are the substances that are used to control the stabilizers.
Fourier Transform Infrared Spectrophotometer:
This instrument is used to identify the raw material, thus used for the identification of drugs.
Procedure:
- Ensure the spectrophotometer is calibrated.
- Ensure the instrument is clean.
- Switch on the main power of the instrument and wait for 5min. to warm up the instrument.
- Ensure before preparation of sample KBr is dried and sample cup, mortal and pastel are clean.
- Don’t use water for cleaning of KBr cell.
- For cleaning of KBr cells use only carbon tetrachloride.
- Scan spectrum as per specification.
- After scanning, select the report format for printing.
- Select the proper printer and take a printout.
- After completion of the analysis put the sample cup or liquid sample holder in the proper place.
- Enter the detail of the analysis in the usage log book.
- Switch off the mains by the end of the day.
- The F.T.I.R. SPECTROPHOTOMETER is manufactured by “JASCO”.
Ultraviolet visual spectrophotometer:
- Ensure the spectrophotometer is calibrated.
- Ensure the instrument is clean.
- Ensure the cuvettes are clean.
- Switch on the instrument then wait for some time until the installation is complete.
- After filling the solution in cuvettes wipe it up with tissue paper outside then put it on the holder.
- Scan and select wavelength as per specification.
- After scanning, select the report format for printing.
- After completion of analysis put the cuvettes in the proper place and keep glassware in the washing room for cleaning.
- Enter the detail of the analysis in the usage log book.
- Switch off the mains by the end of the day.
Viscometer (BROOKFIELD):
- Every liquid has its own flow rate.
- Liquids like water, alcohol, chloroform and acetone move fast, whereas syrup, honey and glycerin flow slowly.
- This rate of flow of liquid depends on the internal resistance involved when one layer moves over another layer.
- In other words the property of a liquid that gives its resistance to flow is called viscosity.
Measurement of Viscosity:
- The viscosity is measured in both C.G.S. and S.I. systems.
- In the C.G.S. system the viscosity of a liquid is measured in dyne seconds per square centimeter.
- It is known as POISE.
- Each poise is further divided into 100 centipoises.
- In the S.I. system the viscosity is measured in Newton per square meter.
- The viscosity of water is one centipoise.
Friabilator(U.S.P.):
- A Friability test is performed to evaluate the ability of the tablet to withstand wear and tear in packing, handling and transporting.
- The apparatus used to perform this test is known as the fabricator.
- The apparatus consists of a plastic chamber, which is divided into two parts and it revolves at a speed of 25rpm.
- Twenty tablets are weighed and placed in the plastic chamber.
- The chamber is rotated for 4 minutes or 100 revolutions.
- During each revolution tablet falls from a distance of 6 inches.
- The tablets are removed from the chamber and weighed.
- Loss in weight indicates friability.
- The tablets are considered to be of good quality if the loss in weight is less than 0.8%.
- Friabilator is manufactured by “ELECTROLAB”
Moisture analyzer:
- The instrument used is Karl Fisher Reagent to detect moisture in the sample.
- Firstly to calibrate the instrument water factor is calculated.
- Then the moisture is found in percentage.
- The moisture analyzer is manufactured by “METLER TOLEDO”.
MICROBIOLOGY LABORATORY
- Microbiology has immense value in a pharmaceutical company.
- All micro-level testing is done in the micro lab.
- Care is taken that no micro-organisms could enter the lab.
Entry procedure to Microbiological Lab:
- Ensure that the visitors shall be accompanied by the Q.C. staff.
- Open the door of the change room of the microbiology lab and enter the change room.
- Remove the apron of the Q.C. lab and placed it in the defined place.
- Sit on the crossover bench and wear the shoe cover.
- Cross the crossover bench.
- Wear the apron and cap provided for the microbiology lab.
- Rinse the hands with provided disinfectant solution (STERLLIUM).
- Enter the microbiology lab.
Exit procedure to Microbiological Lab:
- Enter the change room from the microbiology lab.
- Remove the apron and cap of the microbiology lab and placed them in the defined place.
- Cross the crossover bench.
- Remove the shoe cover and placed it in the defined place.
- Wear the apron of Q.C. lab.
- Exit from the microbiology lab change room.
Autoclave:
- Moist heating is done in an autoclave.
- It consists of a strong metallic chamber usually made of stainless steel.
- It has a cover fitted with a steam vent, a pressure gauge and a safety valve.
- A rubber gasket is fitted on the inner side of the lid, in order to make the autoclave airtight.
- The cover is closed by wing nuts and bolts.
- The electrically heated element is fitted at the bottom to heat the water to convert it into steam.
- The perforated inner chamber is placed on the stand.
- The material to be sterilized is loosely packed into it.
- A sufficient quantity of water is poured into the chamber after removing the perforated chamber.
- The level of water is adjusted in such a way that it does not touch the bottom of the perforated chamber.
- The lid is closed by wing nuts and bolts.
- The autoclave is switched on to heat the water.
- The vent is open and the safety valve is set at the required pressure.
- When steam starts coming out from the vent and it continues for 5 min, it is then closed.
- It indicates that the air has been removed.
- The steam pressure starts rising and it comes to the desired pressure (i.e. 10 lbs/square inch) with the corresponding temperature of 115°c or 15 lbs/square inch.
- After the stated period switch off the autoclave.
- Allow it to cool to about 40°c before opening the vent.
- When the whole of the steam inside the autoclave is removed the lid is opened and the sterilized material is taken out.
- It is manufactured by “OSWORLD”.
Hot-Air Oven:
- It is used for sterilization of pharmaceutical products and other materials.
- It is a double-walled chamber made of steel insulation material, such as glass fibers of asbestos is filled between the two walls of the oven to avoid heat loss.
- The door is also double walled having asbestos filled between two walls and having asbestos gasket on its inner side.
- 2 or 3 perforated shelves are fixed inside to place the material for sterilization.
- An electric fan is also fitted to ensure the uniform circulation of hot air in order to maintain the required temperature on all the shelves.
- Heating elements are fitted on the bottom of the oven and it is thermostatically controlled.
- A thermometer is fitted in the oven to note down the temperature inside the oven.
- After heating the content of the oven for 2 hours at 160°
- The articles are allowed to remain there, till the temperature comes down to 40°
- The sterilized material is then removed from the oven.
- It is manufactured by “THERMOLAB”.
T.O.C. Analyser (Total Organic Carbon Analyser):
- It is used for purifying the water.
- It removes the volatile substance from water.
- This procedure is applicable for sampling and testing of purified water, tablet and capsule, liquid dosage forms and ointment manufacturing facilities.
Pass Box:
- It is used to pass the sterile product to the sampling room.
- It is used to avoid contamination of sterile products.
Digital pH Meter:
- With the pH meter we can determine the pH level of the ingredients.
- It is manufactured by “LAB INDIA”.
Colony counter:
- It is used to determine the percentage of colonies present in fungi and bacteria.
B.O.D. Incubator (Biological Oxygen Demand Incubator):
- In this type of incubator, the fungi test has occurred.
- In between the temperature 20°to 25°c for 72-100 hours.
- It is also used for bacterial tests between the temperature of 30° to 38°c for 48-72 hours.
H.P.H.V. Steam Sterilizer (High-Pressure High Vacuum):
- It involves the following processes:
- Vacuum Leak Test
- Bowie & Dick Test
- Standard Process
- HPHV
- All these processes make the steam sterilizer superior to the autoclave.
- In this type of sterilizer mass volume of media is sterilized at one time.
Hot Plate:
- It is used to heat all types of media, solid and liquid materials.
- Temperature range 0° to 100°
AIR HANDLING UNIT (A.H.U)
- AHU controls the micro-organism, temperature, humidity and air in the industrial environment.
- It develops a suitable working environment for workers.
- AHU consists of four units :
Air filters: Air filters (5micron) and ultrafilters are used for removing dust particles and micro-organisms from the air, so only filtered air entered in the industry.
Temperature controller: It deposes the temperature of incoming air according to need.
Humidifier: It removes the excess humidity from the air inlet which is necessary for preventing the sticking of the capsule and deterioration of hygroscopic substance.
Fresh air blower: Finally fresh air (oxygenated) is injected into the working environment by the fresh air blower.
Conclusion :
- The packaging unit is an integral part of the pharmaceutical industry.
- Any dosage form is likely to get contaminated upon prolonged exposure to environmental conditions.
- As a result, for maintaining purity, integrity and homogeneity of a product.
- It should be packed properly which will not only help in enhancing the shelf life of the product but also will be of importance in assuring the right amount of drug as mentioned in the label that will help people lead healthier lives.
PURIFIED WATER SYSTEM
- Water is used throughout the pharmaceutical industry, as a raw material, a process intermediate, a product ingredient, or as a finished product itself.
- Its wide use in many of applications demands that the manufacturers look very closely at regulatory requirements, specifications, purification methods, installation and validation.
- The water system is always a major area of interest during regulatory inspections and still represents a significant.
- So, in every formulation purified water is used which is according to IP.
- It involves the following components :
- Soft water pipeline
- Heat exchanger
- Cartridge Filter
- Reverse Osmosis
- Ion Exchanger / Electronic Deionization
- Ultrafiltration
- Storage tank
- Ultraviolet
- Thus Purified water obtained is supplied to every department.
- This water is tested daily against microorganisms.
- The design concept will become the industry standard, as more and more pharmaceutical companies employ this elegant and effective solution to realize and appreciate its many benefits, advantages and efficiencies over conventional hygienic plumbing methods.
QUALITY ASSURANCE
- An organization is having inside management rules and these rules are managed by quality assurance.
- In contritely situating, it provides candidacy in the sampler and is an important part of the system in action.
- The availability of the complete system of slandered operating precedence.
- These all actions need to be taken in the standard way, this means that everyone involve in pharmaceutical manufacturing has a back of procedure that find in the way.
- They should do their job and provide a standard way of working.
Responsibilities of QA:
- It must ensure that products are formulated and developed in accordance with QA principle.
- It must provide all job formats of all manufacturing, and testing methods.
- It must insure that there are up-to-date return procedures to control all starting material intermediate and bulk products.
- It must insure that no product is released for distribution without all the documents pertaining to it having been checked by an authorized person.
REFERENCES
- Good manufacturing practices for pharmaceuticals: A plan for total Quality control.2nd edition; Sidndey H. willing, Murray M. Tuckerman, etc.
- Pharmaceutics: Basic Principles and Application to Pharmacy Practice.
- Good laboratory practices and current good manufacturing practices; Ludwing Huber.
- Aulton’s Pharmaceutics by Michael E. Aulton (Editor); Kevin M. G. Taylor.
- Pharmaceutical Dosage Forms and Drug Delivery Systems by Loyd V. Allen
- Martin’s Physical Pharmacy and Pharmaceutical Sciences by Patrick J. Sinko
- Modern Pharmaceutics, Fifth Edition, Volume 1 by Alexander T. Florence. (Editor); Jurgen Siepmann (Editor); Juergen Siepmann.
- Theory and Practice of Contemporary Pharmaceutics by Tapash K. Ghosh (Editor); Bhaskara R. Jasti (Editor); Craig Svensson.

